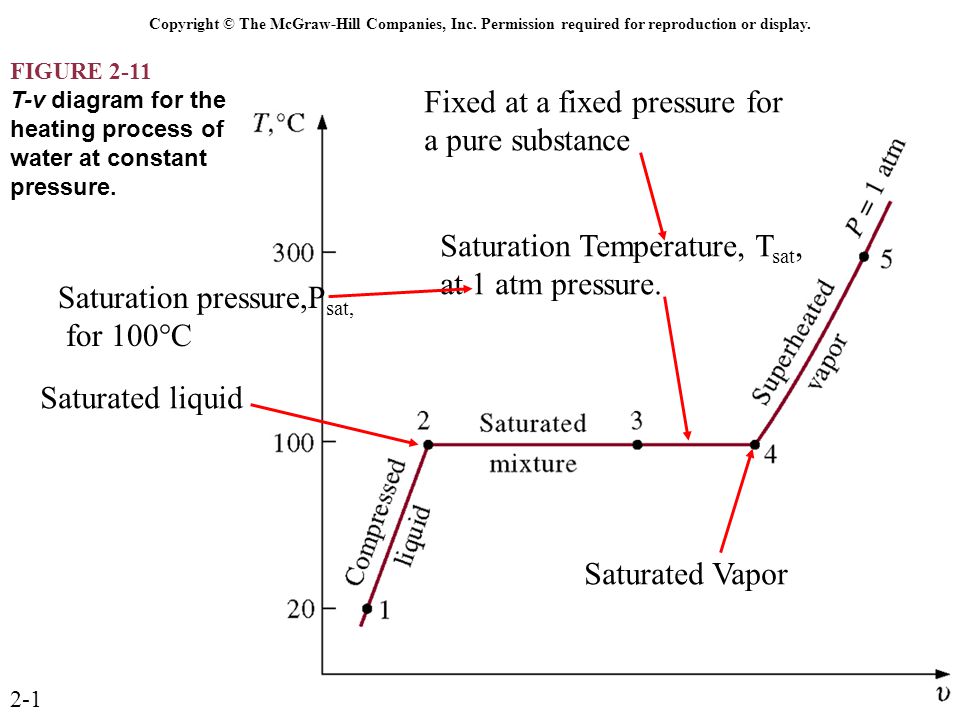
T V Diagram For Pure Substance
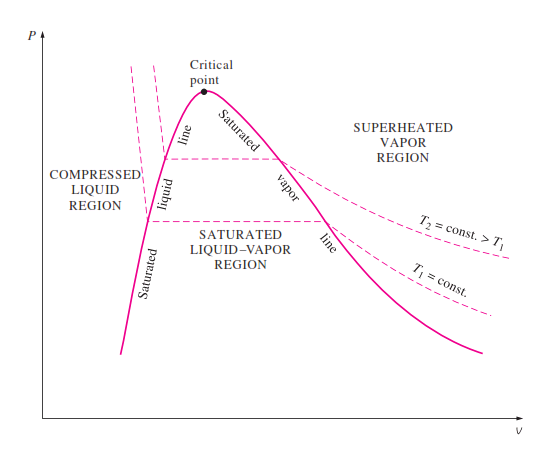
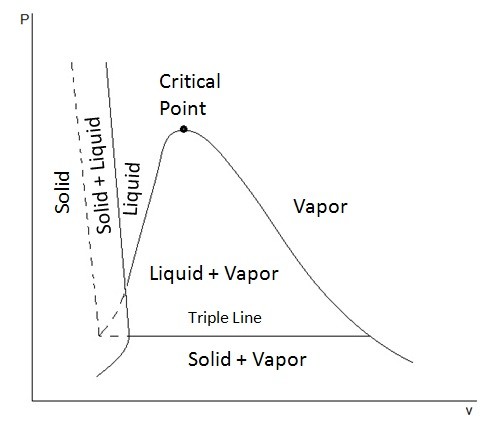
P t v diagram for a substance that contracts during freezing left and for a substance that expends during freezing right.
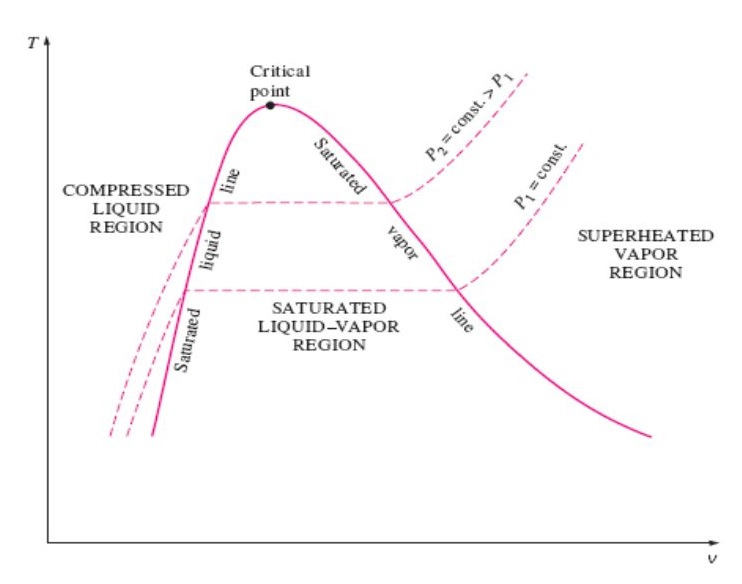
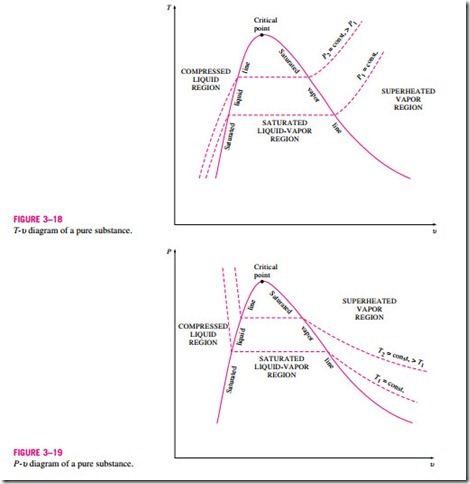
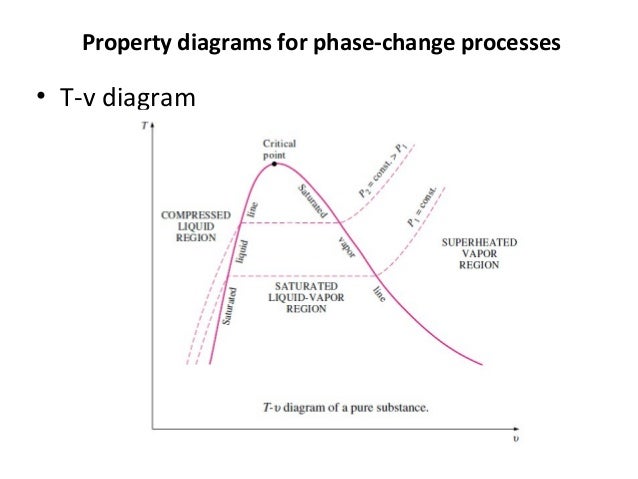
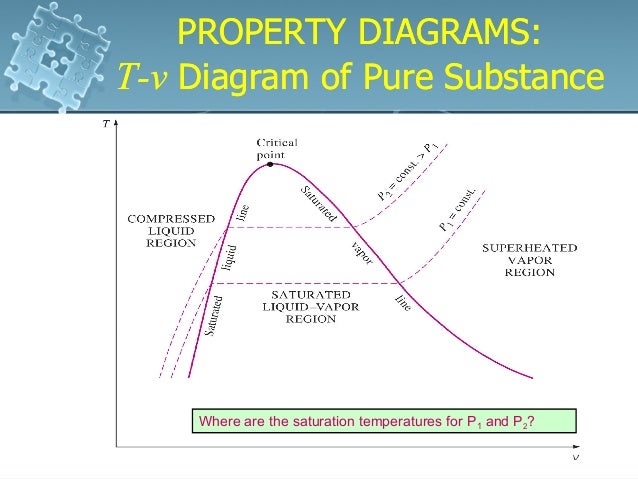
T v diagram for pure substance. In the cases we ll be looking at on this page the phases will simply be the solid liquid or vapour gas states of a pure substance. The plot of p p t v is a surface called p v t surface. 4 points get more help from chegg. Notice that the t v diagram is based exclusively on intensive properties hence mass is not indicated on the diagram.
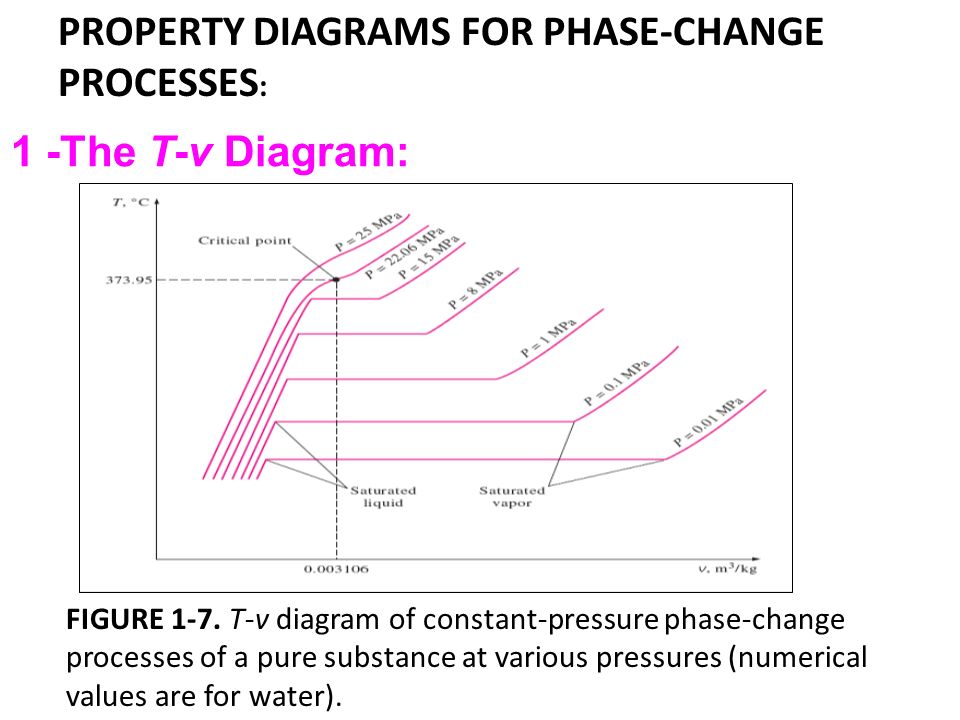
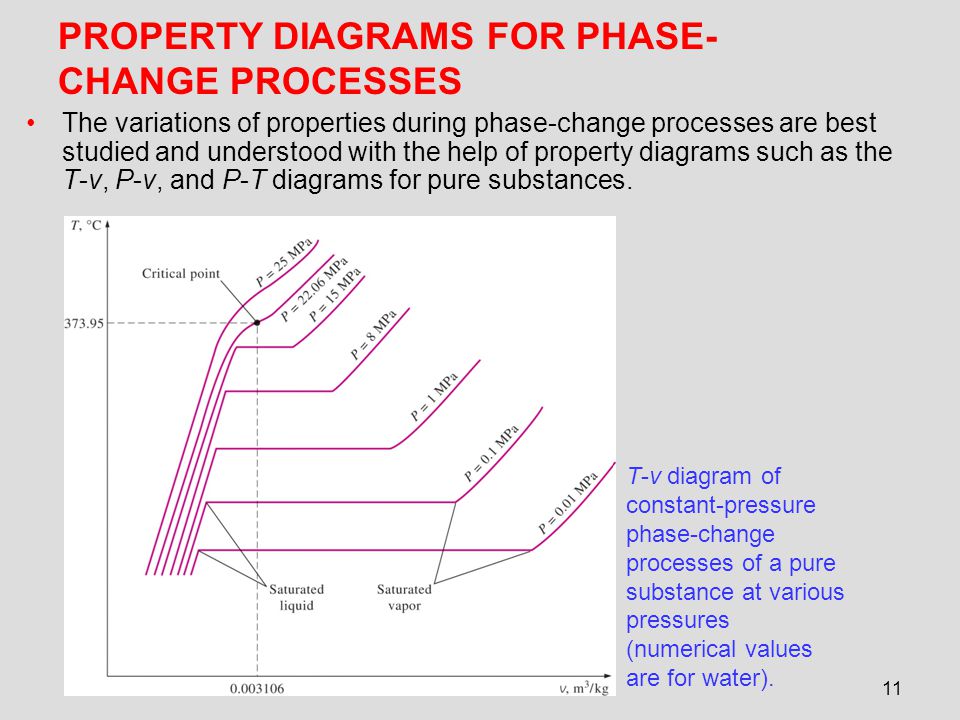
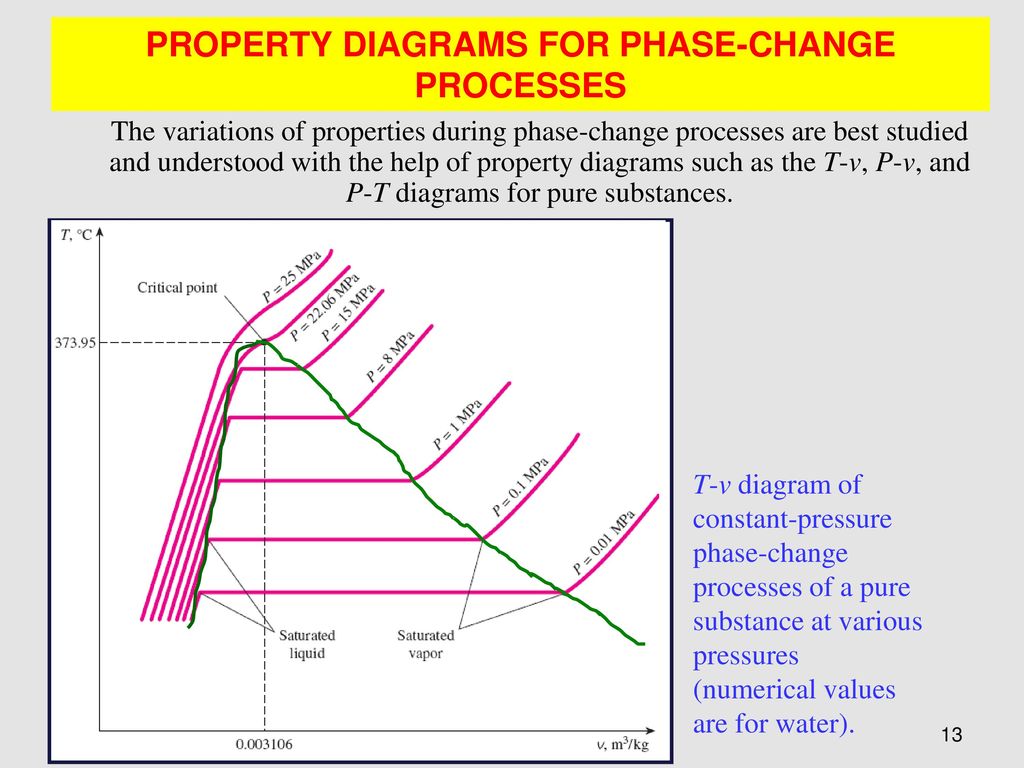
Explain t v diagram of constant pressureb phase change processes of a pure substance at various pressures 0 003106 numerical values are for water. If the pure substance is heated at low pressure until its triple point pressure and temperature at which three phases of a pure substance coexist is reached and while heating the vapor pressure of a solid is measured at various temperatures and then plotted on a p t diagram shown in fig. Label all the curves regions and indicate the location of the critical point. Subscribe subscribed unsubscribe 1 65m.
This is the phase diagram for a typical pure substance. T v diagram for pure substance working fluid i e. What is the enthalpy of vaporization and what is its value at the critical point. Thermodynamics 9 properties of pure substances p t v diagram figure 9.
Pure substances t v diagram tutorials point india ltd. Unsubscribe from tutorials point india ltd. Thus we indicate on the diagram that in order to determine the quality at state 2 we need to first evaluate the specific volume v 2 which can then be compared to the saturation values v f and v g at the pressure of 100 kpa. Please leave any questions you have in the comments section.
Draw a schematic of the t v diagram for a pure substance. This video explains how to draw t v diagrams and what they represent. Property tables in addition to the temperature pressure and specific volume data tables. P p t v.
Figure 3 1 1 p v t surface and projections for a substance that expands on freezing. Pressure can be expressed as a function of temperature and specific volume. We will again secure one another t v curve as shown in figure for pressure of p2 we have taken p2 for the value of pressure 1 mpa. Water is now at pressure of 1 mpa let us consider that we are providing the heat energy to the system at constant pressure of 1 mpa.
The temperature volume diagram for pure substance at different constant pressure is shown below.